
by Julie Matthews, MS | Jan 3, 2024 | BioIndividual Nutrition Articles, Chronic Conditions, Scientific Research
New science and clinical experience reveal concerns about oxalates that far exceed traditional kidney stone pathology. In order to best support their patients and clients, integrative practitioners, and especially diet and nutrition specialists would benefit from...

by Julie Matthews, MS | Sep 22, 2016 | All Posts, Autism & Pediatric Articles, Scientific Research
If you are a nutrition practicing health professional you’ve likely heard of methyl B12. If you’re a patient or this is new to you as a practitioner, you will enjoy learning about this important subject. An exciting study indicates that...

by Julie Matthews, MS | Aug 5, 2016 | Autism & Pediatric Articles, Chronic Conditions, Scientific Research
>> DOWNLOAD and read the PDF of this article In my earlier article about oxalates, I explained how oxalates influence the biochemical progression and symptomatic expression of varied chronic diseases. Through an overarching “lens” of 18 years’ research and...
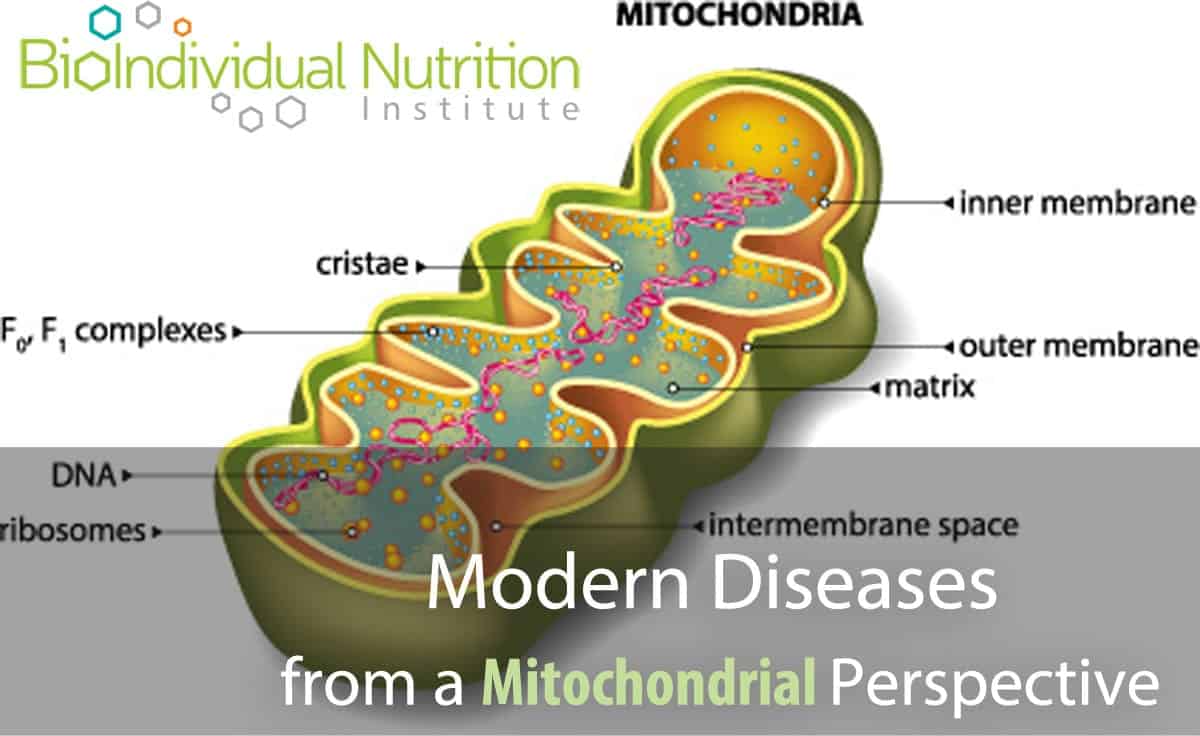
by Julie Matthews, MS | Jul 23, 2016 | BioIndividual Nutrition Articles, Chronic Conditions, Scientific Research
A growing body of research is implicating mitochondrial dysfunction as a root cause of a wide spectrum of metabolic, lifestyle and degenerative diseases including: Chronic Fatigue Syndrome, Autism, Cancer, Diabetes, Alzheimer’s, Fibromyalgia and potentially many more....

by Julie Matthews, MS | Jul 20, 2016 | BioIndividual Nutrition Articles, Scientific Research
I’m thrilled to announce the launch of Kara Fitzgerald and Romilly Hodges’ new book the “Methylation Diet and Lifestyle.” Methylation is a topic we cover extensively in our Advanced Practitioner training program at the BioIndividual Nutrition Institute. I’ve admired...

by Julie Matthews, MS | Apr 1, 2015 | Autism & Pediatric Articles, Scientific Research
Recently, there has been quite a lot of controversy in the media (and even in the nutrition field) on whether diet and nutritional supplements can help autism. I’ve been focused on the science and application of nutrition and special diets for...







